New publication: Compartment‐specific screening tools for persistence – potential role and application in the regulatory context
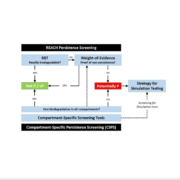
The persistence assessment under REACH relies on compartment‐specific degradation half‐lives derived from laboratory simulation studies with surface water, aquatic sediment or soil. Although these data are given priority, they are not available for most of the compounds. Therefore, according to the integrated assessment and testing strategy for persistence assessment, results from ready biodegradability tests are […]

 T. Junker
T. Junker Mark Woodward
Mark Woodward
 foto-kathrein.de
foto-kathrein.de D. Leib
D. Leib L. Schlüter-Vorberg
L. Schlüter-Vorberg
 SETAC
SETAC D. Leib
D. Leib